Dr. Alexander Seed: "Organic Synthesis: New Heterocyclic Methodology, Synthesis of HNO Donors, Liquid Crystal Synthesis"

| Dr. Alexander Seed Ph.D. University of Hull, UK, 1995 Associate Professor 134 Science Research Building aseed@kent.edu 330-672-9528 |
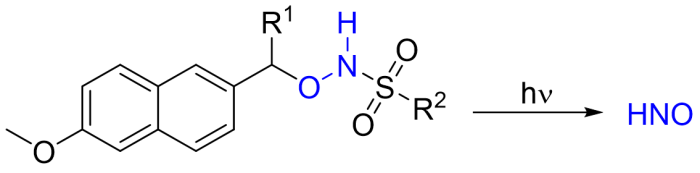
Synthesis of Photoactivatable HNO Donor Molecules
We are synthesizing new organic HNO (nitroxyl) donor molecules which have shown promise for the rapid (sub-second) generation of HNO, a biologically relevant redox sibling of the well-known cell signaling molecule nitric oxide. There is much current interest in nitroxyl, which shows clinically promising activity relating to cardiovascular health. Since HNO rapidly dimerizes in aqueous solution, its study requires the use of HNO donor molecules. Most known HNO donors decompose to release nitroxyl slowly (~minutes-to-hours), often under non-physiological conditions. Consequently, there is an urgent need for HNO donors which more rapidly and cleanly generate HNO with spatial and temporal control. We are synthesizing HNO donors (e.g., 1 and related compounds) which will rapidly generate HNO “on demand” via photolytic cleavage of an O-protecting group followed by fast HNO elimination. In collaboration with colleagues in New Zealand, we are studying the kinetics of their photodecomposition and the kinetics and mechanisms of the reactions between HNO and various biomolecules.
Synthesis of Ferroelectric Liquid Crystals Based on Novel S-Heterocyclic and Fluorinated S-Heterocyclic Cores (collaboration with Paul Sampson)
Our group is exploring the development of new synthetic methodology and approaches to the construction of a variety of novel thiophene, thieno[2,3-b and 3,2-b]thiophene, 1,3-thiazole and 1,3,4-thiadiazole ring systems decorated with alkoxy- and/or fluoro- substituents. These building blocks are utilized in the synthesis of new ferroelectric and high birefringence liquid crystalline materials (e.g., compounds 2-6). One of the long-term goals of our program is to develop a detailed understanding of the structure-mesophase properties of a family of ferroelectric liquid crystals that, in many cases, exhibit the chiral smectic C phase without the appearance of unwanted chevron defects. Physical studies will allow us to elucidate whether these are DeVries materials or if there is another effect that explains the unusual lack of chevron defects seen upon cooling into the SmC phase.
![Our group is exploring the development of new synthetic methodology and approaches to the construction of a variety of novel thiophene, thieno[2,3-b and 3,2-b]thiophene, 1,3-thiazole and 1,3,4-thiadiazole ring systems decorated with alkoxy- and/or fluoro-substituents.](https://www-s3-live.kent.edu/s3fs-root/s3fs-public/styles/fixed_width_700/public/seed-research-002.png?VersionId=G6.VhGkRZ6KooWlBMAOyFjViZdTytekc&itok=gY7FK5Rf)
Relevant Publications
- R.B. Cink, Y. Zhou, L. Du, M.S. Rahman, D.L. Phillips, M. Cather Simpson, A.J. Seed, P. Sampson and N.E. Brasch, Mechanistic Insights into Rapid Generation of Nitroxyl from a Photocaged N‑Hydroxysulfonamide Incorporating the (6-Hydroxynaphthalen-2-yl)methyl Chromophore, J. Org. Chem., 2021, 86, pp. 8056-8068.
- Y. Zhou, R.B. Cink, A.J. Seed, M.C. Simpson, N.E. Brasch, and P. Sampson, Stoichiometric Nitroxyl (HNO) Photorelease using the (6-Hydroxy-2-naphthalenyl)methyl Phototrigger, Org. Lett., 2019, 21(4), pp. 1054-1057.
- A.J. Seed and Paul Sampson, A review of self-organizing 2,5- and 2,4-disubstituted 1,3-thiazole-containing materials: Synthesis, mechanisms, and tactics, Liq. Cryst., 2017, 44(12-13), pp. 1894-1910.
- Zhou, Y.; Cink, R.B.; Dassanayake, R.S.; Seed, A.J.; Brasch, N.E.; Sampson, P. Rapid Photoactivated Generation of Nitroxyl (HNO) under Neutral pH Conditions. Angew. Chem. Int. Ed., 2016, 55(42), pp. 13229-13232.
- J.I. Tietz, A.J. Seed, and P. Sampson, Preparation of brominated 2-alkoxythiophenes via oxidation and etherification of 2-thienyltrifluoroborate salts. Org. Lett., 2012, 14(19), pp. 5058-5061.
- A.J. Seed, J.I. Tietz, R.M. Gipson, Y. Yu, and P. Sampson, Low Molar Mass Thieno[3,2-b] and Thieno[2,3-b]thiophenes in Liquid Crystal Materials Science: Recent Synthetic Approaches, Liq. Cryst., 2015, 42(5-6), pp. 918-927.
- A. Seed, Synthesis of self-organizing mesogenic materials containing a sulfur-based five-membered heterocyclic core, Chem. Soc. Rev., 2007, 36(12), pp. 2046-2069.
- A.A. Kiryanov, P. Sampson and A.J. Seed, Synthesis of 2-alkoxy-substituted thiophenes, 1,3-thiazoles and related S-heterocycles via Lawesson’s reagent-mediated cyclization under microwave irradiation: applications for liquid crystal synthesis, J. Org. Chem., 2001, 66(23), pp. 7925-7929.
- C. Zhang, A.M. Grubb, A.J. Seed, P. Sampson, A Jákli, and O.D. Lavrentovich, Nanostructure of edge dislocations in a smectic-C liquid crystal, Phys. Rev. Lett., 2015, 115 (8), article no. 08780.
- J.I. Tietz, P. Sampson, and A.J. Seed, Novel 5-(4-alkoxyphenyl)thieno[3,2-b]thiophene-2- carboxylate esters: Highly efficient synthesis and mesogenic evaluation of a new class of materials exhibiting the SmC phase, Liq. Cryst., 2012, 39(5), pp. 515-530.
- A.M. Grubb, C. Zhang, A. Jákli, P. Sampson, and A.J. Seed, 2-Alkoxythiazoles: A new core unit for incorporation into self-organizing materials. Synthetic approach, mesomorphism, and electrooptic evaluation. Liq. Cryst., 2012, 39(10), pp. 1175-1195.
- A.M. Grubb, S. Hasan, A.A. Kiryanov, P. Sampson, and A.J. Seed, The synthesis and physical evaluation of 5-alkoxy-1,3-thiazoles prepared via Lawesson’s reagent-mediated cyclisation of -benzamido esters, Liq. Cryst., 2009, 36(5), pp. 443-453.
- R.M. Gipson, P. Sampson, and A.J. Seed, The synthesis and mesogenic behavior of the first series of low molar mass thieno[3,2-b]thienothiophene-2-carboxylate ester-based mesogens, Liq. Cryst., 2010, 37(1), pp. 101-108.
- A.M. Grubb, M.J. Schmidt, A.J. Seed, and Paul Sampson, Convenient preparation of halo-1,3-thiazoles: Important building blocks for materials and pharmaceutical synthesis, Synthesis, 2012, 44(7), pp. 1026-1029.
- Brian Sybo, Patrick Bradley, Alan Grubb, Seth Miller, Katie Proctor, Lucy Clowes, M. Ruth Lawrie, Paul Sampson and Alexander J. Seed, 1,3,4-Thiadiazole-2-carboxylate esters: New synthetic methodology for the preparation of an elusive family of self-organizing materials, J. Mater. Chem., 2007, 17(32), pp. 3406-3411
- B. K. McCoy, Z. Q. Liu, S. T. Wang, R. Pindak, K. Takekoshi, K. Ema, A. Seed, and C. C. Huang, Smectic-C* phase with two coexistent helical pitch values and a first-order Smectic-C* to Smectic-C* transition, Phys Rev. E., 2007, 75(5-1), article no. 051706.
- A.A. Kiryanov, A.J. Seed and P. Sampson, Ring fluorinated thiophenes: applications to liquid crystal synthesis, Tetrahedron Lett., 2001, 42(50), pp. 8797-8800.
- V.M. Sonpatki, M.R. Herbert, L.M. Sandvoss and A.J. Seed, Troublesome alkoxythiophenes. A highly efficient synthesis via cyclization of -keto esters, J. Org. Chem., 2001, 66(22), pp. 7283-7286.
