


Opportunities for Undergraduate Research
Independent research is an important and rewarding part of the undergraduate chemistry experience.
Many students begin by enrolling in Individual Investigation (CHEM 40796). Some choose to expand their work into a senior thesis project. Both options give you the opportunity to work one-on-one with a faculty mentor and develop the skills needed to succeed as a practicing chemist.
Research allows you to build hands-on experience, strengthen your critical thinking, and explore questions that go beyond the traditional lab classroom. Undergraduate researchers often present their work at professional conferences and may even publish in scientific journals. These experiences can strengthen applications to graduate programs, medical or professional schools, and industry positions.
The Department of Chemistry and Biochemistry offers a wide range of research opportunities for undergraduates. On this page, you’ll find brief descriptions of faculty research interests and information about those who are currently accepting undergraduate researchers.
If a faculty member’s work interests you, reach out to them directly to express your interest. Once you’ve identified a faculty mentor who is willing to supervise your project, you can register for the appropriate course and begin your research journey.
For questions, please contact the Undergraduate Office at (330) 672-2405 or chemug@kent.edu.
Undergraduate Research Projects
-
Dr. Sanjaya Abeysirigunawardena: "Modified nucleotides in ribosomal RNA and modification enzymes influence ribosomal assembly"

Dr. Sanjaya Abeysirigunawardena
Ph.D. Wayne State University, 2008
Associate Professor
126 Integrated Sciences Building
sabeysir@kent.edu
330-672-2667
Study of Epitranscriptomic Regulation (funded by National Institutes of Health)
Understand the evolutionary importance of modified nucleotides in RNA-protein interactions during epitranscriptomic regulation.
- Investigate the key structural and sequence properties of proteins important for m6A recognition using phage display methods.
- Discover novel methyl-readers those involved in epitranscriptomic regulation using protein pulldown assays followed by bioinformatic analysis.
- Investigate key differences between direct and indirect readings of m6 A.
Drug Discovery
Discovery of small molecules that bind to various RNA drug targets.
- Discovery of novel antibacterial and anticancer agents by inhibiting RNA modification enzymes.
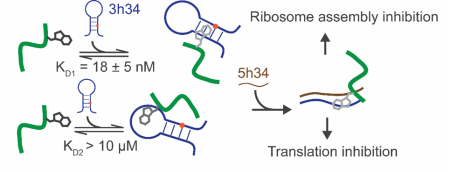
Ribosome Biogenesis
Investigate the ability of modification enzymes and their respective modifications to influence bacterial ribosomal assembly.
- Study the impact of modification enzymes on the co-transcriptional assembly of bacterial 30S ribosome subunit.
- Investigation of the ability of modification enzymes to modulate RNA dynamics using single molecule spectroscopy.
- Determination of the correlation of the antibiotic resistance in bacteria, ribosome assembly, and modification enzymes.
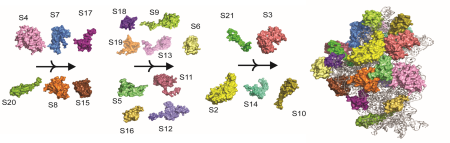
Meet the People in the Abey Research Group
Five graduate and over 30 undergraduate researchers have been actively involved in research in the Abey group. These undergraduate researchers include students majoring in Chemistry, Biology, and Biotechnology. In the Abey Lab, we strive for inclusivity!

To learn more about the Abey Research Group, please visit our website.
-
Dr. Scott Bunge: "Inorganic and Materials Chemistry"

Dr. Scott Bunge
Ph.D. Georgia Institute of Technology, 2001
Associate Professor
311E Williams Hall
sbunge@kent.edu
330-672-3816
Inorganic and Materials Chemistry
Inorganic and Materials Chemistry:
Our research group intends to make a strong contribution to the fields of inorganic chemistry, nanoscale science and technology, and materials science. With this aim, standard Schlenk and glovebox techniques are employed to synthesize a variety of low-coordinate air and moisture sensitive inorganic precursors. Characterization methods include multinuclear NMR, X-ray crystallography, FT-IR and UV/VIS spectroscopy, TGA/DTA, SEM, TEM and X-ray powder diffraction.
These investigations, while rooted in traditional aspects of chemistry, will often involve students in collaborations with an array of other scientists and engineers. Group members will have their own projects; however, each group member's research will have significant overlap with others in the group. As such, the students' depth of fundamental chemical principles will become augmented by exposure to a breadth of additional concepts. It is anticipated that such a combination of skills results in a fertile and creative environment for achievement of research goals. Therefore, students should frequently expand beyond the reaches of classical chemistry subjects, and embrace additional areas, as required, for the successful execution of a specific project.
Self-Assembly of Inorganic Nanocrystals:
It is envisioned that working on nanoscale materials will lead to unprecedented products in electronics, biotechnology, medicine, transportation, agriculture, environment, national security and other fields. To achieve these products, one of our initial goals is attaining a fundamental scientific understanding of nanoscale phenomena, particularly collective phenomena. In support of this goal our research proposes a rational and convenient method to construct and examine the properties of hybrid, self-assembled inorganic/organic nanostructures. A variety of individuals (from 1st year undergraduate students to experienced postdoctoral fellows) will contribute to this project while gaining both training and education in nanotechnology. This is a fundamental tenant of the nation’s nanotechnology initiative.
Gold Chemistry:
In today's society, gold chemistry currently has an important role in fields such as electronics and medicine. However, there is still a current lack of understanding in the fundamental reaction chemistry of gold. The development of gold (I) chemistry is dominated by the viewpoint that gold is a prototypical soft Lewis acid, which forms its most stable complexes with soft Lewis bases. Accordingly, the synthesis of gold (I) complexes with hard Lewis bases such as oxygen, nitrogen, or carbon has been limited to a select number of examples. Such complexes have been described as intrinsically unstable, and therefore, have a pronounced tendency to either decompose to gold metal or aggregate into ill-defined clusters. Similar problems, although to a lesser extent, have been described for copper and silver. This instability has historically been described as a limitation to the development of gold chemistry. However, a few recent reports have hinted that a much richer field of coordination chemistry might be accessible. Therefore, in order to contribute to the understanding of this important metal, it is the goal of this research proposal to investigate the chemistry of previously "inaccessible" gold (I) alkyls, amides and alkoxides.
These novel complexes will be isolated as crystalline solids and characterized via solution and solid-state NMR, X-ray crystallograpy, FT-IR and UV/VIS spectroscopy. Throughout this investigation, the stability and reaction chemistry of these complexes will be investigated. It is the intention of this proposal to generate a large family of complexes in order to gain a true appreciation for the intrinsic stability of gold complexes
Group 11 Metal-Organic Precursors:
The semiconductor industry continues to undergo rapid technological changes, especially in fabricating nanoscale integrated circuit (IC) devices. Smaller device features and a need for increased chip surface area have led to the use of multilevel interconnections to increase the functionality of IC devices. The search for better performance has led to consideration of materials such as Cu, Ag, and AU for use as interconnections. Historically, industry has relied on well-established approaches, such as physical vapor deposition, to create such interconnects. However, due to considerations such as cost and the nanoscale size regime the devices now have entered, interconnects fabricated via metal-organic chemical vapor deposition (MOCVD) and more recently nanocrystal deposition are increasingly favored. Currently, there is a lack of suitable CU, Ag, and Au precursors that have attributes desired for MOCVD and nanocrystal synthesis.
-
Dr. Bansidhar Datta: "Suppression of Tumor Growth Via Gene Transfer"

Dr. Bansidhar Datta
Ph.D. University of Nebraska, 1989
Associate Professor
014 Science Research Lab
bdatta@kent.edu
330-672-3304
My research focus is to suppress tumor growth in ex-vivo cell culture model and in vivo animal model by regulating the expression of p67 gene. P67, also known as MetAP2, regulates the level of phosphorylation of eukaryotic initiation factor 2 (eIF2) and extracellular signal-regulated kinases 1 and 2 (ERK1/2). EIF2 maintains the rate of global protein synthesis, whereas ERK1/2 controls the cell signaling mediated by the proto-oncogene Ras. In more than 60% of human cancers, Ras is mutated. Expression of mutant Ras in ex-vivo cell culture system transforms cells and injection of these transformed cells into nude mice causes localized tumor formation. However, this tumor growth could be suppressed by overexpressing p67 prior to the injection into nude mice. Well-developed blood vessels were identified in mouse tumors caused due to the expression of mutant Ras whereas, undeveloped and spongy type of blood vessels were more prominent in tumors if p67 was overexpressed in Ras-transformed cells prior to the injection into nude mice. These observations also indicate that p67 is interfering in angiogenesis in tumors.
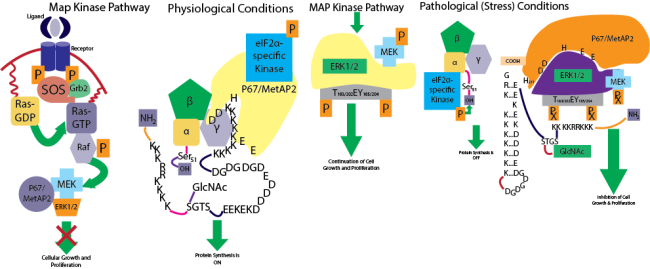
Expression of p67 could be controlled via several mechanisms: (i) when cells were treated with fumagillin, an epoxy compound isolated from fungus, that binds to H231 of p67 covalently and inhibits it auto-proteolytic activity. As a result, p67 accumulates inside the cells and binds to ERK1/2 to inhibit their phosphorylation. This also leads to the dissociation of p67 from EIF2, allowing it to be phosphorylated by its inhibitory kinases and shuts down the global protein synthesis. The inhibition of ERK1/2 phosphorylation by p67 leads to the inhibition of cell signaling mediated by Ras and thus acts as a negative regulator of cell cycle. Together, p67 acts as a mediator communicating with cell cycle and protein synthesis machinery. (ii) The promoter regions of p67 contains several cis-elements that could act as binding sites for different activators, which can control the expression of p67 in response to several growth conditions. (iii) Overexpression of cloned p67 gene in mammalian cells via different expression vectors. Using these different mechanisms we are now focusing on the suppression of cell growth of different human tumor cells.
Scholarly, Creative & Professional Activities
- Datta, B. (2014) Diversified roles of p67/MetAP2 as a regulator of cell growth and differentiation, in tumor suppression, and in obesity (Review). Curr. Topics Biochem. Res. 16:41-52.
- Datta, B., Datta, R., and Tammali, R. (2014) Lysine-rich domains of eukaryotic initiation factor 2-associated glycoprotein, p67 are involved in the suppression of phosphorylation of several cellular kinases. Intl. J. of Biotech. Biochem. 10:141-156.
- Tammali, R., Datta, R., Datta, B. (2014) Phosphorylation of double-stranded RNA activated kinase, PKR is induced by eukaryotic initiation factor 5 (eIF5), which is also a major substrate for this kinase in vitro. Intl. J. of Biotech. Biochem. 10:127-140.
- Datta, B. and Datta, R. (2014) Inhibition of phosphorylation of cdk2 and cdc2 in rat tumor hepatoma cells constitutively expressing specific p67 mutants. Intl. J. of Biotech. Biochem. 10:113-125.
- Datta, B. and Datta, R. (2014) Phosphorylation of eukaryotic initiation factor 2a during differentiation of mouse myoblasts into myotubes is mediated by an unknown kinase. Intl. J. Adv. Res. Chem. Sci. 1:1-5.
- Ghosh,A., Tammali, R., Balusu, R., Datta, R., Chattopadhyay, A., Bhattacharya, M., and Datta, B. (2014) Oligomerization of the eukaryotic initiation factor 2-associated glycoprotein p67 requires N-terminal 1-107 amino acid residues. Intl. J. Appl. Biotech. Biochem. 4:25-44.
- Datta, B., Earl, D., Roods, M., Datta, S. (2014) Analysis of p67/MetAP2 gene from mammals. Intl. J. Mol. Genet. 5:1-12.
- Majumdar, A., Ghosh, A., Datta, S., Prudner, B., and Datta, B. (2010) P67/MetAP2 suppresses K-RasV12 mediated transformation of NIH3T3 mouse fibroblasts in culture and in athymic mice. Biochemistry 49:10146-10157.
- Datta, B., (2009) Roles of P67/MetAP2 as a tumor suppressor – a review. BBA - Reviews on Cancer. 1796:281-292.
- Datta, B., Ghosh, A., Majumdar, A. & Datta, R. (2007) Autoproteolysis of Rat p67 Generates Several Peptide Fragments: The N-Terminal Fragment, p26, Is Required for the Protection of eIF2a from Phosphorylation. Biochemistry 46:3465-3475.
- Ghosh, A., Datta, R., Majumdar, A., Bhattacharya, M., and Datta, B. (2006) The N-terminal lysine residue-rich domain II and the 340-430 amino acid segment of eukaryotic initiation factor 2-associated glycoprotein p67 are the binding sites for the g-subunit of eIF2. Exp. Cell Res. 312:3184-3203.
- Datta, B., Datta, R., Ghosh, A., and Majumdar, A. (2006) The binding between p67 and eukaryotic initiation factor 2 plays important roles in the protection of eIF2alpha from phosphorylation by kinases. Arch. Biochem. Biophys. 452:138-148.
-
Dr. Barry Dunietz: "Quantum Chemistry - Electronic Structure Modeling"

Dr. Barry Dunietz
Ph.D. Columbia University, 2000
Professor
300 Williams Hall
bdunietz@kent.edu
330-672-8401
We employ and develop electronic structure computational tools and molecular models to investigate complex systems. We are mainly interested in improving understanding of structure-function relationships of molecular systems that are involved in transfer or transport of excitation energy or charge carriers. The investigated systems are analyzed in the context of optoelectronic applications as in photovoltaics and in the context of photosynthesis in natural systems.
Our studies are pursued in collaboration with related synthesis, spectroscopy and device fabrication experimental efforts. In addition we collaborate with theoreticians and computational experts of complimentary expertise to synergize our research program.
Our group activity involves members that are high school, undergraduate, and graduate students, and postgraduate fellows. We are all engaged in highly collaborative efforts extending both within the group and with external experts.
We pursue state-of-the-art density functional theory-based models to study energy and electron transport properties of extended molecular systems. Our efforts are funded by several agencies. Further details can be obtained at our Research Group Website or by contacting us directly.
Publications
- A Comparative Study of Different Methods for Calculating Electronic Transition Rates. Alexei A. Kananenka, Xiang Sun, Alexander Schubert, Barry D. Dunietz, and Eitan Geva, J. Chem. Phys., 148(2018), 102304.
- Enhancing Charge Mobilities in Organic Semiconductors by Selective Fluorination: A Design Approach Based on a Quantum Mechanical Perspective. B. Maiti, A. Schubert, S. Sarkar, S. Bhandari, et. al., Chem. Sci., 8(2017), 6947-6953.
- Phosphorescence in Bromobenzaldehyde Can Be Enhanced Through Intramolecular Heavy Atom Effect. S. Sarkar, H. P. Hendrickson, D. Lee, F. DeVine, J. Jung, E. Geva, J. Kim, and B. D. Dunietz, J. Phys. Chem. C., 121(2017), 3771-3777.
- Achieving Predictive Description of Molecular Conductance by Using a Range-Separated Hybrid Functional. Atsushi Yamada, Qingguo Feng, Austin Hoskins, Kevin D. Fenk, and Barry D. Dunietz, Nano Lett.,16 (2016), 6092-6098.
- Deleterious Effects of Exact Exchange Functionals on Predictions of Molecular Conductance. Qingguo Feng, Atsushi Yamada, Roi Baer, and Barry D. Dunietz, J. Chem. Theory Comput., 12(2016), 3431-3435.
- The Effect of Interfacial Geometry on Charge-Transfer States in the Phthalocyanine/Fullerene Organic Photovoltaic System. Myeong H. Lee, Eitan Geva, and Barry D. Dunietz, J. Phys. Chem. A., 12(2016), 2970-2975.
-
Dr. Mahinda Gangoda: "Analytical Instrumentation Facility Research Opportunities"

Dr. Mahinda Gangoda
Ph.D. Kent State University, 1983
Research Engineer
017 Williams Hall
mgangoda@kent.edu
330-687-4157
Research Opportunities in the Analytical Instrumentation Facility
The Analytical Instrumentation Facility (AIF), located in the Department of Chemistry and Biochemistry, performs pilot or short-term industrial research projects using a wide range of state-of-the-art analytical instruments.
The major instruments and instrumental techniques used in these research projects are: NMR, GC, GC/MS, HPLC, HPLC/MS, DSC, TGA, elemental analysis, FT-IR and UV-VS. Interested students will receive training on the instrumentation if they have no previous experience with it.
-
Dr. Torsten Hegmann: "Liquid Crystal - Nanomaterial Interactions"

Dr. Torsten Hegmann
Ph.D. Martin Luther University, Halle (Germany), 2001
Professor
201E Liquid Crystals & Materials Science Building
thegmann@kent.edu
330-672-7770
Research in our group focuses on three major themes: (1) Understanding interactions in nanoparticle-liquid crystal dispersions, (2) Synthesis, self-assembly and properties of anisometric nanomaterials, and (3) Nanomaterials for biological and medical applications (incl. cell-specific uptake and brain drug delivery). All research areas are highly multidisciplinary and collaborative. Students and postdocs working on these themes develop skills in organic as well as nanomaterial synthesis, a wide range of materials characterization techniques, and to some degree soft matter physics, pharmacology, biological sciences as well as medical research. We have key collaborations with experts in their respective fields in place, which is critical when tackling multidisciplinary research targets.
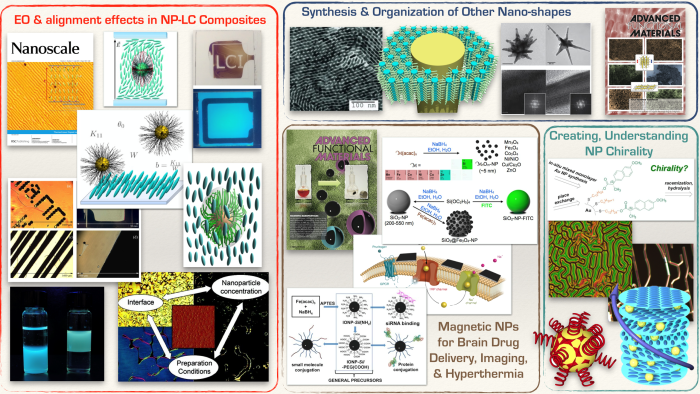
Recent Publications
- T. Hegmann et al., Liq. Cryst. 2015, in press. “Chemically and thermally stable, emissive carbon dots as viable alternatives to semiconductor quantum dots for emissive nematic liquid crystal–nanoparticle mixtures with lower threshold voltage”
- T. Hegmann et al., J. Mater. Chem. B 2015, 3, 6877. “Aqueous synthesis of polyhedral “brick-like” iron oxide nanoparticles for hyperthermia and T2 MRI contrast enhancement”
- T. Hegmann, W. Haase et al., RSC Advances 2015, 5, 34491. “Electro-optic and dielectric properties of a ferroelectric liquid crystal doped with chemically and thermally stable, emissive carbon dots”
- T. Hegmann et al., Adv. Funct. Mater. 2015, 25, 1180. “Discotic liquid crystal functionalized gold nanorods: 2- and 3D self-assembly plus macroscopic alignment and increased charge carrier mobility in hexagonal columnar liquid crystal hosts affected by molecular packing and π-π interactions”.
- T. Hegmann et al., ACS Nano 2014, 8, 11966. “Detecting, visualizing, and measuring gold nanoparticle chirality using helical pitch measurements in nematic liquid crystal phases”
- T. Hegmann, D. W. Miller et al., Int. J. Nanomed. 2014, 9, 3013. "Magnetic field enhanced convective diffusion of iron oxide nanoparticles in an osmotically disrupted cell culture model of the blood-brain barrier"
- T. Hegmann, W. Haase et al., Chem. Phys. Lett. 2014, 599, 80. “Influence of different amount of functionalized bulky gold nanorods dopant on the electrooptical, dielectric and optical properties of the FLC host”
- T. Hegmann, H.-S. Kitzerow et al., ChemPhysChem 2014, 15, 1395. “Nanoparticle doping in nematic liquid crystals: Distinction between surface and bulk effects by numerical simulations”
- H.-S. Kitzerow, T. Hegmann et al., ChemPhysChem 2014, 15, 1381. “Synthesis of liquid crystal silane functionalized gold nanoparticles and their effects on optical and electro-optic properties of a structurally related nematic liquid crystal”
- T. Hegmann et al., Part. Part. Syst. Charact. 2014, 31, 257. "Ink-jet printed nanoparticle alignment layers: Easy design and fabrication of patterned alignment layers for nematic liquid crystals"
- T. Hegmann et al., Langmuir 2013, 29, 10850. "One-pot synthesis of iron oxide nanoparticles with functional silane shell: A versatile general precursor for conjugations and biomedical applications"
-
Dr. Songping Huang: "Nanomedicine – Development of Novel Nanoparticles to Target the Vulnerability of Iron Metabolism for Treating Bacterial Infections"

Dr. Songping Huang
Ph.D. Michigan State University, 1993
Professor
330 Integrated Sciences Building
shuang1@kent.edu
330-672-2230
Research work in Huang Group is divided into three major areas: (1) rational design and synthesis of small-molecule antimicrobial agents that have different cellular or molecular targets than those of the conventional antibiotics for combating antimicrobial resistance (AMR); (2) harnessing cytotoxicity from ferroptosis for cancer treatment; and (3) development of MRI contrast agents for gastrointestinal (GI) tract imaging.
Antibiotics have underpinned modern medicine for well over 70 years. Their use has reduced childhood mortality, increased life expectancy, made invasive surgery and organ transplant possible, and ensured the safety of cancer chemotherapy, to name just a few. Meanwhile, in response to the widespread use and misuse of antibiotics in humans and animals, bacterial pathogens have accelerated the process of mutation to develop drug resistance to these medicines. As a result, drug-resistant bacterial pathogens have started to spread faster than the discovery of new antibiotics. It is estimated that more than 30% of clinical isolates of Pseudomonas aeruginosa (P. aeruginosa) from the patients in any given intensive care unit (ICU) or a nursing home are now resistant to three or more antibiotic drugs. The situation is very similar for other pathogenic organisms. It is alarming to note that some strains have become resistant to virtually all the commonly available antimicrobial agents.
We focus on the discovery of new molecules that can act as effective NDH-2 inhibitors to overcome ARM. NDH-2, also known as type II dehydrogenase, is a peripheral membrane protein functioning as an enzyme (EC: 1.6.99.3) to catalyze the electron transfer in respiratory chain of certain pathogenic organisms e.g., Staphylococcus aureus (SA), but not expressed in mammals, hence constituting an ideal target for rational drug design. Since NDH-2 is not a known target of any existing antimicrobial drugs, our drug lead compound 6c exhibits not only unusual potency against SA, but also the remarkable ability to overcome the drug resistance in a number of antibiotic-resistant SA mutant bacteria. Currently, 6c is under preclinical animal studies for development as a topical antimicrobial drug to treat skin and soft-tissue MRSA (i.e., methicillin-resistant staphylococcus aureus) infections, particularly those by the MRSA mutant bacteria that are resistant to mupirocin and fusidic acid, the two widely used topical antibiotics for treating skin and soft-tissue MRSA infections.
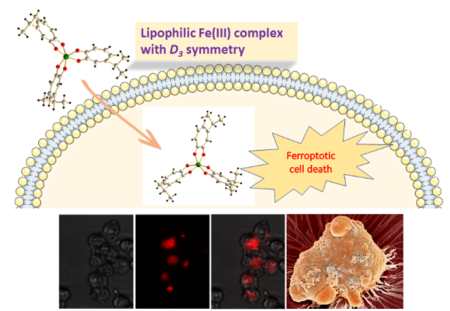
Ferroptosis is a special type of programmed cell death triggered by cellular dysregulation of iron. We capitalize on ferroptosis as a new mode of action to develop Fe-based metallodrugs for cancer chemotherapy with the aim to improve the selectivity of cytotoxicity in cancerous cells vs. normal cells to reduce toxic side effects of anticancer drugs. Iron is known as a double-edged sword in biology: it is essential for almost all forms of life for their survival, division and growth, yet iron is potentially cytotoxic as dysregulated cellular iron uptake can result in reactive oxygen species (ROS)-triggered cell death from the iron-catalyzed Fenton reaction. Through the judicial selection of lipophilic iron chelators with high affinity for Fe(III) as the ionophores, we have demonstrated that certain octahedral Fe(III) complexes with D3 symmetry have the remarkable ability to transport iron across the cell membrane to trigger intracellular Fenton reaction, which affords potent broad-spectrum in vitro anticancer activity against a plethora of cancer cell lines including the Pt-resistant ovarian cancer cell line. Since the cellular targets and the mode of action of such Fe(III) complexes are drastically different and nonoverlapping with those of cisplatin, they have the distinct ability to overcome Pt resistance in A2780cis ovarian cancer cells. Our work in this area raises the possibility of harnessing ferroptosis to develop Fe-based metallodrugs for cancer chemotherapy.
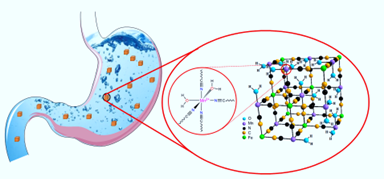
The current commercial contrast agents (CAs) used in MRI are small-molecule Gd3+-chelates that do not have sufficient stability in acidic environment of the stomach and temporal stability when they move through the GI tract, making the development of oral MRI contrast agents based on the platform of Gd3+-complexes problematic. Our group pioneered the use of biocompatible NPs of Prussian blue (PB) and its many analogues as oral contrast agents for MRI applications. Such NPs exhibit extremely high stability in the gastric juice, high r1 relaxivity, low toxicity and high temporal stability, showing great potential for the development of true T1-weighted oral contrast agentsfor MR imaging of the entire GI tract, an unmet clinical need in diagnostic medicine.
Selected Publications:
- N. Abeydeera, B. Yu, P. D. Bishnu, M. Kim, S. D. Huang “Harnessing the Toxicity of Dysregulated Iron Uptake for Killing Staphylococcus aureus: Reality or Mirage?” Biomaterials Science (IF=6.843),10, 474-484 (2022). https://doi.org/10.1039/D1BM01743H
- Z. Wang, J. Li, B. M. Benin, B. Yu, S. D. Bunge, N. Abeydeera, S. D. Huang, M.-H. Kim “Lipophilic Ga Complex with Broad-Spectrum Antimicrobial Activity and the Ability to Overcome Gallium Resistance in both Pseudomonas aeruginosa and Staphylococcus aureus” Journal Medicinal Chemistry (IF=7.446), 64: 9381-9388 (2021). https://doi.org/10.1021/acs.jmedchem.1c00656
- M. S. Kandanapitiye, T. M. Dassanayake, A. C. Dassanayake, J. Shelestak, R. J. Clements, S. D. Huang “K2Mn3[FeII(CN)6]2 NPs with High T1-Relaxivity Attributable to Water Coordination on the Mn(II) Center for Gastrointestinal Tract MR Imaging” Advanced. Healthcare Materials (IF=9.933), 10(20): 2100987 (2021). https://doi.org/10.1002/adhm.202100987
- Dyne E, Prakash P, Li J, Yu B, Schmidt T, Huang S, Kim MH. “Mild Magnetic Nanoparticle Hyperthermia Promotes the Disaggregation and Microglia-mediated Clearance of Beta-Amyloid Plaques” Nanomedicine: Nanotechnology, Biology, and Medicine (IF=6.458), 34:102397 (2021). doi.org/10.1016/j.nano.2021.102397; https://pubmed.ncbi.nlm.nih.gov/33857686
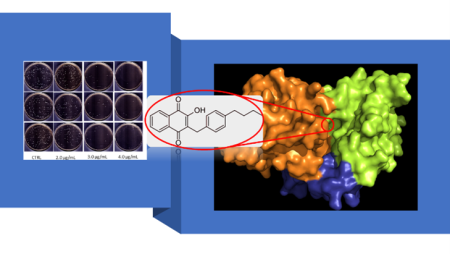
- Song R, Yu B, Friedrich D, Li J, Shen H, Krautscheid H, Huang SD, Kim MH. “Napthoquinone-derivative as a synthetic compound to overcome the antibiotic resistance of methicillin-resistant S. aureus” Communications Biology (IF=6.268), 3(1):529 (2020). https://www.nature.com/articles/s42003-020-01261-0
- Yu B., Wang Z., Almutairi L., Huang S., Kim MH “Harnessing iron oxide nanoparticles towards the improved bactericidal activity of macrophages against Staphylococcus aureus” Nanomedicine: Nanotechnology, Biology, and Medicine (IF=6.458), 24:102158 (2020). https://pubmed.ncbi.nlm.nih.gov/31982615; https://onlinelibrary.wiley.com/doi/abs/10.1002/ange.201713177
- Wang Z, Yu B, Alamri H, Yarabarla S, Kim MH, Huang SD “KCa(H2O)2[FeIII(CN)6].H2O nanoparticles as a novel antimicrobial agent for Staphylococcus aureus” Angewandte Chemie Intd ((IF=15.334).) 37:2214-2218 (2018). https://onlinelibrary.wiley.com/doi/abs/10.1002/ange.201713177
-
Dr. Arkaprabha Konar: "Ultrafast Photochemistry and Photophysics of Biochemical systems"

Dr. Arkaprabha Konar
Ph.D. Michigan State University, 2015
Assistant Professor
114 Science Research Building
akonar@kent.edu
330-672-2034
Ultrafast Photochemistry and Photophysics of Biochemical Systems
Konar research group aims to use the techniques of nonlinear multidimensional spectroscopy and microscopy to unravel the dynamics of ultrafast processes (femtoseconds – nanoseconds) such as energy and charge transfer in biological systems over a diverse range of time and length scales as well as in new materials used for energy conversion and conservation. We are in the phase of setting up the first state of the art ultrafast laser lab at Kent State University and you could be a part of this exciting process and get hands on training on working with ultrafast lasers, optical alignment, building light sources in different frequency ranges among other things.
Selected Publications
- Y. Song, A. Konar, R. Sechrist, V. P. Roy, R. Duan, J. Dziurgot, V. Policht, K. J. Kubarych and J. P. Ogilvie, “Multispectral Multidimensional Spectrometer Spanning the Ultraviolet to the Mid-Infrared”, Rev. Sci. Inst., 2019, 90, 013108
- T. E. Wiley, A. Konar, N. A. Miller, K. G. Spears and R. J. Sension, “Primed and Ready for Action: Ultrafast Excited State Dynamics and Optical Manipulation of a Four Stage Rotary Molecular Motor”, J. Phys. Chem. A, 2018, 122, 7548
- A. Konar, R. Sechrist, Y. Song, V. Policht, P. D. Laible, D. F. Bocian, D. Holten, C. Kirmaier and J. P. Ogilvie, “Electronic Interactions in the Bacterial Reaction Center revealed by Two-Color 2D Electronic Spectroscopy”, J. Phys. Chem. Lett., 2018, 9, 5219
- A. Konar, Y. Shu, V. V. Lozovoy, J. E. Jackson, B. G. Levine and M. Dantus, “Polyatomic Molecules under Intense Femtosecond Laser Irradiation”, J. Phys. Chem. A, 2014, 118, 11433
-
Dr. Hanbin Mao: "Mechano-analytical Chemistry: A New Interdisciplinary Field"

Dr. Hanbin Mao
Ph.D. Texas A&M University, 2003
Professor
038 Science Research Building
hmao@kent.edu
330-672-9380
Mechano-analytical Chemistry: A New Interdisciplinary Field
By combining Analytical Chemistry and Single-molecule Biophysics, we created a new chemistry field: Mechano-Analytical Chemistry. We have used the following techniques to develop this new field:
Lab-on-a-chip
Micro total analysis system (mTAS), a.k.a. lab-on-a-chip, integrates a variety of lab components on a chip as small as one inch square in area. In a typical lab-on-a-chip scheme, chemicals can be synthesized, purified and analyzed on a single chip. The technique is indispensable in the emerging fields such as genomics and proteomics, where huge sets of data are collected and analyzed. It is also very useful in the screening processes to identify promising drug leads or optimal conditions for crystallization. The low cost and high-throughput capability make this technique ideal for sensor development. Combining with laser tweezers or magnetic tweezers (see below), our lab is interested in the methodology development and bioanalytical application of this technique, for example in the fields of ultra sensitive sensors and screening methods.
Laser Tweezers
Since the discovery of the laser tweezers in the 1980s, the application of this technique has been mostly limited to the physics where it was originated. The lag of the application in chemistry can be attributed to the following reasons. First, tiny amount of the material contained inside a trapped object prevents the use of many traditional detection methods, such as UV-vis and IR. Second, to build a strong optical trap, objectives with short working distance are often used. This leaves little room to incorporate other detection methods. Our lab uses unique capabilities of the laser tweezers, i.e., force detection in the range of piconewtons and spatial measurement down to Angstroms, to follow the chemical interactions such as binding events between receptors and ligands.
Magnetic Tweezers
Compared to laser tweezers (see above), magnetic tweezers have advantages of lower force range (fN–pN), less drift, and full compatibility with a typical lab-on-a-chip layout. Most importantly, magnetic tweezers have high-throughput. Magnetic objects can be easily incorporated into microfluidic channels on a chip and manipulated by an external magnet. These objects can be used to control the fluidics at the micrometer scale, which is one of the most difficult tasks in the development of lab-on-a-chip techniques.
Using laser- and magnetic-tweezers on a chip, we have been investigating fundamental properties of biomacromolecules including DNA and proteins. In addition, we have pioneered ultrasensitive and high-throughput biosensing method called Single-Molecule Mechanochemical Sensing (or SMMS). With many patents awarded, we have used SMMS to detect trace level of biomarkers for various diseases, as well as toxins such as mercury in the environment.
Our research is interdisciplinary. We carry out many collaborative projects with groups from materials and biosciences. Incoming members have ample opportunities to learn subjects through coworkers from other fields.
Selected Publications
- "A Single-Molecule Platform for Investigation of Interactions between G-quadruplexes and Small-Molecule Ligands". Deepak Koirala, Soma Dhakal, Beth Ashbridge, Yuta Sannohe, Raphine Rodriguez, Hiroshi Sugiyama, Shankar Balasubramanian, Hanbin Mao, Nature Chemistry, 2011, 3, 782-787.
- "Interaction of G-quadruplexes in the Full-length 3' Human Telomeric Overhang", Jibin Abraham Punnoose, Yunxi Cui, Deepak Koirala, Philip M. Yangyuoru, Chiran Ghimire, Prakash Shrestha, and Hanbin Mao. Journal of the American Chemical Society, 2014,136 (52), 18062-18069.
-
Dr. Marianne Prévôt: "Printing the Next Generation of Sustainable Plastic Optics Across Multiple Dimensions and Length Scales"

Dr. Marianne Prévôt
Ph.D. University of Rennes, France, 2015
Assistant Professor
093 Integrated Sciences Building
mprevot1@kent.edu
330-672-3166
Our group explores the potential and viability of biodegradable, environmentally friendly, luminescent bioplastic materials. Our research, which focuses on the functionalization of nanomaterials and polymer materials with liquid crystal moieties, has the potential to revolutionize diverse optical uses in medicine, sensing, and information.
Emissive Functional Liquid Crystal Devices
This research focuses on understanding the efficiency of chirality transfer (also called amplification) across length scales, with the objective of controlling it for the next generation of products in polarized optics. We design circularly polarized emissive materials with tunable properties, actuatable through various stimuli such as heat, light, electric and magnetic fields.
We utilize a liquid crystal strategy to design ultra-efficient circularly polarized luminescent devices with unique and enhanced specifications. These devices can be envisioned as chiroptical probes and provide higher sensitivity and resolution in chemical and biological optical sensing. They can also be used as chiroptical switches for encrypted integrated logic devices with higher storage density and security. These optical elements feature high accuracy with low cost through rapid prototyping. The long-term goal of this research is to progress its practical relevance from a static element to a dynamic optical device allowing to advance the real-world applications of the “smart” materials.
Zero-Power Liquid Crystal-based Sensors
The main objective of this project is to develop diverse production approaches to deploy zero-power, highly customizable, lightweight, and low-cost sensors that can detect and monitor acute and chronic exposures to a multitude of toxic gases. The key goals are to develop sensors that provide (i) high sensor responsivity, (ii) high sensor sensitivity and selectivity, and (iii) stability in time. The portable, mountable, zero-power gas detectors are capable of detecting a variety of toxic gases within the same platform with significantly improved specificity and selectivity. The sensor functionality will dramatically assist in faster assessing a hazardous environment. The research will ultimately participate in creating a safe environment for the defense personnel and first responders.
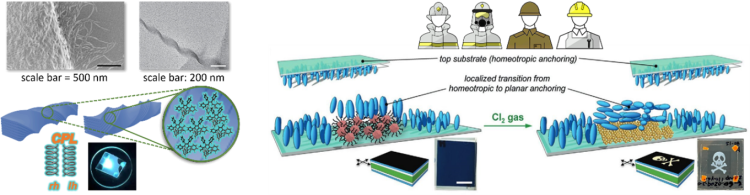
The Prévôt Lab focuses on printing the next generation of sustainable plastic optics across multiple dimensions and industries. (Left) Highly tunable circularly polarized emission of an aggregation-induced emission dye using liquid crystal-based helical nano- and microfilaments as supramolecular chiral templates. (Right) Zero-power optical, ppt- to ppm-level toxic gas and vapor sensors with image, text, and analytical capabilities.
Selected publications:
- B. Sezgin, J. Liu, D.P.N. Gonçalves, C. Zhu, T. Tilki, M.E. Prévôt*, T. Hegmann*, “Controlling the structure and morphology of organic nanofilaments using external stimuli”, ACS Nanoscience Au, 2023, 3, 295-309, DOI:10.1021/acsnanoscienceau.3c00005.
- M.E. Prévôt, S. Ustunel, G. Freychet, C.R. Webb, M. Zhernenkov, R. Pindak, R.J. Clements, E. Hegmann, “Physical models from physical templates using biocompatible liquid crystal elastomers as morphologically programmable inks for 3D printing”, Macromolecular Bioscience, 2023, 23, 2200343, DOI:10.1002/mabi.202200343.
- J. Liu, Y. Molard, M.E. Prévôt*, T. Hegmann*, “Highly tunable circularly polarized emission of an aggregation-induced emission dye using helical nano- and microfilaments as supramolecular chiral templates”, ACS Applied Materials & Interfaces, 2022, 14, 25, 29398, DOI: 10.1021/acsami.2c05012.
- M.E. Prévôt, S. Ustunel, B. Yavitt, G. Freychet, C.R. Webb, M. Zhernenkov, E. Hegmann, and R. Pindak, “Synchrotron microbeam diffraction studies on the alignment within 3D-printed smectic-A liquid crystal elastomer filaments during extrusion”, Crystals, 2021, 11, 5, 523, DOI:10.3390/cryst11050523.
- J. Liu, S. Shadpour, M.E. Prévôt, M. Chirgwin, A. Nemati, E. Hegmann, R.P. Lemieux, and T. Hegmann, “Molecular conformation of bent-core molecules affected by chiral side chains dictates polymorphism and chirality in organic nano- and microfilaments”, ACS Nano, 2021, 15, 4, 7249, DOI:10.1021/acsnano.1c00527.
- S. Ustunel#, M.E. Prévôt#, G.A.R. Rohaley, C.R. Webb, B. Yavitt, G. Freychet, M. Zhernenkov, R. Pindak, E. Schaible, C. Zhu, T. Hegmann, R.J. Clements, and E. Hegmann, “Mechanically tunable Elastomer and Cellulose Nanocrystal composites as Scaffolds for In vitro Cell Studies”, Materials Advances, 2020, 2, 1, 464, DOI: 10.1039/D0MA00676A.
- M.E. Prévôt, A. Nemati, T.R. Cull, E. Hegmann, and T. Hegmann, “A zero-power optical, ppt- to ppm-level toxic gas and vapor sensor with image, text, and analytical capabilities”, Advanced Materials Technologies, 2020, 2000058, DOI: 10.1002/admt.202000058.
-
Dr. Alexander Seed: "Organic Synthesis: New Heterocyclic Methodology, Synthesis of HNO Donors, Liquid Crystal Synthesis"

Dr. Alexander Seed
Ph.D. University of Hull, UK, 1995
Associate Professor
134 Science Research Building
aseed@kent.edu
330-672-9528
Synthesis of Photoactivatable HNO Donor Molecules
We are synthesizing new organic HNO (nitroxyl) donor molecules which have shown promise for the rapid (sub-second) generation of HNO, a biologically relevant redox sibling of the well-known cell signaling molecule nitric oxide. There is much current interest in nitroxyl, which shows clinically promising activity relating to cardiovascular health. Since HNO rapidly dimerizes in aqueous solution, its study requires the use of HNO donor molecules. Most known HNO donors decompose to release nitroxyl slowly (~minutes-to-hours), often under non-physiological conditions. Consequently, there is an urgent need for HNO donors which more rapidly and cleanly generate HNO with spatial and temporal control. We are synthesizing HNO donors (e.g., 1 and related compounds) which will rapidly generate HNO “on demand” via photolytic cleavage of an O-protecting group followed by fast HNO elimination. In collaboration with colleagues in New Zealand, we are studying the kinetics of their photodecomposition and the kinetics and mechanisms of the reactions between HNO and various biomolecules.
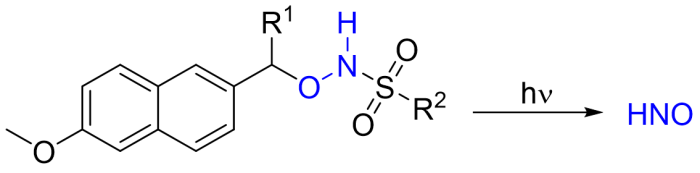
Synthesis of Ferroelectric Liquid Crystals Based on Novel S-Heterocyclic and Fluorinated S-Heterocyclic Cores (collaboration with Paul Sampson)
Our group is exploring the development of new synthetic methodology and approaches to the construction of a variety of novel thiophene, thieno[2,3-b and 3,2-b]thiophene, 1,3-thiazole and 1,3,4-thiadiazole ring systems decorated with alkoxy- and/or fluoro- substituents. These building blocks are utilized in the synthesis of new ferroelectric and high birefringence liquid crystalline materials (e.g., compounds 2-6). One of the long-term goals of our program is to develop a detailed understanding of the structure-mesophase properties of a family of ferroelectric liquid crystals that, in many cases, exhibit the chiral smectic C phase without the appearance of unwanted chevron defects. Physical studies will allow us to elucidate whether these are DeVries materials or if there is another effect that explains the unusual lack of chevron defects seen upon cooling into the SmC phase.
![Our group is exploring the development of new synthetic methodology and approaches to the construction of a variety of novel thiophene, thieno[2,3-b and 3,2-b]thiophene, 1,3-thiazole and 1,3,4-thiadiazole ring systems decorated with alkoxy- and/or fluoro-substituents.](https://www-s3-live.kent.edu/s3fs-root/s3fs-public/styles/fixed_width_700/public/seed-research-002.png?VersionId=G6.VhGkRZ6KooWlBMAOyFjViZdTytekc&itok=gY7FK5Rf)
Relevant Publications
- R.B. Cink, Y. Zhou, L. Du, M.S. Rahman, D.L. Phillips, M. Cather Simpson, A.J. Seed, P. Sampson and N.E. Brasch, Mechanistic Insights into Rapid Generation of Nitroxyl from a Photocaged N‑Hydroxysulfonamide Incorporating the (6-Hydroxynaphthalen-2-yl)methyl Chromophore, J. Org. Chem., 2021, 86, pp. 8056-8068.
- Y. Zhou, R.B. Cink, A.J. Seed, M.C. Simpson, N.E. Brasch, and P. Sampson, Stoichiometric Nitroxyl (HNO) Photorelease using the (6-Hydroxy-2-naphthalenyl)methyl Phototrigger, Org. Lett., 2019, 21(4), pp. 1054-1057.
- A.J. Seed and Paul Sampson, A review of self-organizing 2,5- and 2,4-disubstituted 1,3-thiazole-containing materials: Synthesis, mechanisms, and tactics, Liq. Cryst., 2017, 44(12-13), pp. 1894-1910.
- Zhou, Y.; Cink, R.B.; Dassanayake, R.S.; Seed, A.J.; Brasch, N.E.; Sampson, P. Rapid Photoactivated Generation of Nitroxyl (HNO) under Neutral pH Conditions. Angew. Chem. Int. Ed., 2016, 55(42), pp. 13229-13232.
- J.I. Tietz, A.J. Seed, and P. Sampson, Preparation of brominated 2-alkoxythiophenes via oxidation and etherification of 2-thienyltrifluoroborate salts. Org. Lett., 2012, 14(19), pp. 5058-5061.
- A.J. Seed, J.I. Tietz, R.M. Gipson, Y. Yu, and P. Sampson, Low Molar Mass Thieno[3,2-b] and Thieno[2,3-b]thiophenes in Liquid Crystal Materials Science: Recent Synthetic Approaches, Liq. Cryst., 2015, 42(5-6), pp. 918-927.
- A. Seed, Synthesis of self-organizing mesogenic materials containing a sulfur-based five-membered heterocyclic core, Chem. Soc. Rev., 2007, 36(12), pp. 2046-2069.
- A.A. Kiryanov, P. Sampson and A.J. Seed, Synthesis of 2-alkoxy-substituted thiophenes, 1,3-thiazoles and related S-heterocycles via Lawesson’s reagent-mediated cyclization under microwave irradiation: applications for liquid crystal synthesis, J. Org. Chem., 2001, 66(23), pp. 7925-7929.
- C. Zhang, A.M. Grubb, A.J. Seed, P. Sampson, A Jákli, and O.D. Lavrentovich, Nanostructure of edge dislocations in a smectic-C liquid crystal, Phys. Rev. Lett., 2015, 115 (8), article no. 08780.
- J.I. Tietz, P. Sampson, and A.J. Seed, Novel 5-(4-alkoxyphenyl)thieno[3,2-b]thiophene-2- carboxylate esters: Highly efficient synthesis and mesogenic evaluation of a new class of materials exhibiting the SmC phase, Liq. Cryst., 2012, 39(5), pp. 515-530.
- A.M. Grubb, C. Zhang, A. Jákli, P. Sampson, and A.J. Seed, 2-Alkoxythiazoles: A new core unit for incorporation into self-organizing materials. Synthetic approach, mesomorphism, and electrooptic evaluation. Liq. Cryst., 2012, 39(10), pp. 1175-1195.
- A.M. Grubb, S. Hasan, A.A. Kiryanov, P. Sampson, and A.J. Seed, The synthesis and physical evaluation of 5-alkoxy-1,3-thiazoles prepared via Lawesson’s reagent-mediated cyclisation of -benzamido esters, Liq. Cryst., 2009, 36(5), pp. 443-453.
- R.M. Gipson, P. Sampson, and A.J. Seed, The synthesis and mesogenic behavior of the first series of low molar mass thieno[3,2-b]thienothiophene-2-carboxylate ester-based mesogens, Liq. Cryst., 2010, 37(1), pp. 101-108.
- A.M. Grubb, M.J. Schmidt, A.J. Seed, and Paul Sampson, Convenient preparation of halo-1,3-thiazoles: Important building blocks for materials and pharmaceutical synthesis, Synthesis, 2012, 44(7), pp. 1026-1029.
- Brian Sybo, Patrick Bradley, Alan Grubb, Seth Miller, Katie Proctor, Lucy Clowes, M. Ruth Lawrie, Paul Sampson and Alexander J. Seed, 1,3,4-Thiadiazole-2-carboxylate esters: New synthetic methodology for the preparation of an elusive family of self-organizing materials, J. Mater. Chem., 2007, 17(32), pp. 3406-3411
- B. K. McCoy, Z. Q. Liu, S. T. Wang, R. Pindak, K. Takekoshi, K. Ema, A. Seed, and C. C. Huang, Smectic-C* phase with two coexistent helical pitch values and a first-order Smectic-C* to Smectic-C* transition, Phys Rev. E., 2007, 75(5-1), article no. 051706.
- A.A. Kiryanov, A.J. Seed and P. Sampson, Ring fluorinated thiophenes: applications to liquid crystal synthesis, Tetrahedron Lett., 2001, 42(50), pp. 8797-8800.
- V.M. Sonpatki, M.R. Herbert, L.M. Sandvoss and A.J. Seed, Troublesome alkoxythiophenes. A highly efficient synthesis via cyclization of -keto esters, J. Org. Chem., 2001, 66(22), pp. 7283-7286.
-
Dr. Hao Shen: "Single-Molecule Polymer Chemistry"

Dr. Hao Shen
Ph.D. Cornell University, 2014
Associate Professor
303 Williams Hall
hshen7@kent.edu
330-672-2523
The Shen group focuses on utilizing state-of-the-art single-molecule spectroscopy to investigate the properties of polymeric materials and their potential applications in catalysis and bioseparation. Unlike conventional ensemble measurements, single-molecule spectroscopy analyzes one probing molecule at a time to reveal the performances dictating subpopulations within a system. Its subdiffraction limited localization enables the microscopic to mesoscopic mapping. Moreover, the real-time observation under millisecond resolution enables the dynamic study of complex processes. With this main approach, the Shen group aims to study the following.
Developing advanced microscopy approaches
Thanks to the developments of fluorescence microscopy, researchers nowadays have a non-destructive approach to observe many soft matters and biological systems in-situ. The persistent efforts in the past two decades have broken the diffraction limits, allowing an optical microscope to achieve nanometer precisions. We now understand many systems better than ever. However, the needs for more capable observation tools will never stop. The Shen group will use phase modulations to develop novel imaging methods that enable higher precision and wider observation ranges.
Spatially resolved polymerization kinetics
The world polymer production surpassed 300 megatons in 2015 and is still expanding rapidly. However, a detailed description of the polymerization kinetics lacks because there are some “anomalous” behaviors associated with the reaction types, such as auto-acceleration, radical trapping, gelation, and diffusion induced termination. Such anomalous processes usually result in severe polydispersity in chain sizes within the bulk polymer materials, ultimately downgrading the performances of functional polymers. The Shen group strives to understand how micro-environment affects the polymerization kinetics. The goal is to understand the polymerization mechanisms better and design new strategies to prepare more homogeneous polymers.
Synthetic polymer membranes for protein separations
Proteins carry out vital physiological activities that are widely used in the pharmaceutical and food industries. Each protein exhibits unique physiological functionalities requiring high purity and concentrations when used for manufacturing highly specific products. Chromatography utilizing synthetic polymers as the stationary phase materials is the industrial standard in downstream processing, which may account for as much as 50% of the overall cost to bring a protein product to the market. Consequently, it is critical to understand the dynamics of protein-synthetic polymer interactions, because any such insights will in turn benefit the designing of high-efficient, anti-fouling stationary phase materials to reduce the manufacturing costs for protein-based products. The Shen group uses single-molecule spectroscopy to map out the protein-polymer interactions in real-time.
Selected Publications
- H. Shen, L.J. Tauzin, R. Baiyasi, W. Wang, N.A. Moringo, B. Shuang, C.F. Landes, “Single Particle Tracking: From Theory to Biophysical Applications”, Chemical Reviews, 2017, 117, 7331-7376.
- H. Shen, L. Tauzin, W. Wang, B. Hoener, B. Shuang, L. Kisley, A. Hoggard, C.F. Landes, “Single-Molecule Kinetics of Protein Adsorption on Thin Nylon 6,6 Films”, Analytical Chemistry, 2016, 88, 9926-9933.
- H. Shen, X. Zhou, N. Zou, P. Chen, “Single-Molecule Kinetics Reveals a Hidden Surface Reaction Intermediate in Single-Nanoparticle Catalysis”, Journal of Physical Chemistry C, 2014, 118, 26902-26911.
-
Dr. Michael Tubergen: "High Resolution Microwave Spectroscopy"

Dr. Michael Tubergen
Ph.D. University of Chicago
Professor
035 Science Research Building
mtuberge@kent.edu
330-672-7079
Our research is focused on understanding the role of weakly bonding interactions in determining the conformational structures of large molecules and molecular complexes. Hydrogen bonding, dipole-dipole, and dispersion forces have long been known to determine the structures of molecular complexes formed in the ultracold environment of supersonic expansions; these same forces also preferentially stabilize some molecular conformations over others.
Microwave spectroscopy is an excellent tool for examining these weak interactions. We use a Fourier-transform microwave spectrometer that was recently constructed in our laboratory. Sample is admitted into the spectrometer by a supersonic expansion of argon which both cools the molecules to ~5 K (rotational temperature) and isolates molecules from neighbors—eliminating the possibility that interactions between molecules might distort the conformational structure. Molecular complexes may also be formed in the expansion by entraining two different sample molecules into the carrier gas.
The spectrometer employs two mirrors for microwave frequencies, and they are positioned to establish a resonant cavity (an integral number of wavelengths between the mirrors). The sample is introduced between the mirrors and irradiated with microwave radiation of some known frequency. If the sample absorbs the radiation, it induces an oscillating signal in an antenna mounted on the opposite mirror. The frequency of the signal is equal to the frequency of the rotational transition; a Fourier-transform converts the oscillating signal into a spectrum as a function of frequency. By measuring rotational transitions, we are able to determine molecular moments of inertia and ultimately determine the bond distances and angles.
One area of application is the study of small biological molecules such as amino acids and peptide derivatives. The conformational structures of individual amino acids in a protein collectively determine the three-dimensional structure of the protein and, ultimately, its biological function. We have recently investigated the spectroscopy of a number of amino amides (amide derivatives of amino acids) including alaninamide, prolinamide, and valinamide. We have also collaborated with colleagues at the National Institute of Standards and Technology to record the first rotationally resolved spectra of a linear dipeptide analogue, N-acetyl alanine methyl amide. Data from the rotational spectra of these species are used to generate the most precise and detailed experimental structures.
Interactions between molecules can be studied using the same technique. We have been interested in how hydrogen bonds influence the structures of molecular complexes by measuring the lengths of the hydrogen bonds and examining the dynamics of the internal motions. These projects are interesting because hydrogen bonding is a fundamental interaction in biochemistry. But we would like to extend our previous studies to consider how the formation of an intermolecular hydrogen bond in a molecular complex affects the relative stability of different conformers. Our recent studies of the conformations of alaninamide and alaninamide-water have confirmed a decade-old prediction for a similar system that the most stable structure of the complex will be based on the lowest energy monomer conformation. Recently we have shown that formation of the 1:1 complex 2-aminoethanol-water changes the conformational structure of 2-aminoethanol. The 2-aminoethanol O–C–C–N dihedral angle increases from 58° to 71° upon formation of an intermolecular hydrogen bond.
These studies provide an important link between molecular modeling, which predicts conformational structures of isolated molecules, and biochemistry, which is concerned with structures in solvent environments. Because computer modeling of molecular structures has become widespread in the design of new polymers and drugs, these projects are particularly relevant.
Selected Publications
- Tubergen, M. J. et al. Rotational spectra, nuclear quadrupole hyperfine tensors, and conformational structures of the mustard gas simulent 2-chloroethyl ethyl sulfide. Journal of Molecular Spectroscopy 233, 180-188 (2005).
-
Dr. Zhiqiang Molly Wang

Dr. Zhiqiang Molly Wang
Ph.D. Fudan University, 1999
Professor
138 Science Research Building
zwang3@kent.edu
330-672-3352
Our research interests in a broad sense are to understand the biological functions of certain proteins from their structural point of view. Particularly, we focus on structure-function relationships of metalloenzymes, as well as protein-protein interaction and protein-ligand interactions that are related to signal transductions in cells. Our long-term goal is to translate these basic research studies into the discovery of new drugs and therapeutic methods to treat human diseases.
Our research interests are in the following areas:
- Regulation of Nitric Oxide Synthesis in Bacterial Nitric Oxide Synthase (NOS)
- Cellular Metabolic Pathway in Pulmonary Arterial Hypertension (PAH)
- Develop Copper Chelating Nanoparticles for PAH and Cancer
- Receptor and Protein Interactions in NO Signaling
- Structure-function Studies of Heme O2 and NO Sensors
- Mechanism of NOS-related Endothelial Dysfunction
Selected Publications:
- Ding, Y., Sun, D., Wang, G., Yu H., Meng, S., Chen, J., Xie, Y., Wang, Z.Q. “In vivo study of doxorubicin-loaded cell penetrating peptide-modified pH-sensitive liposomes: biocompatibility, bio-distribution, and pharmacodynamics in BALB/C-nude mice bearing human breast tumors ", Drug Design, Development and Therapy, 11, 1–13, 2017.
- Wang, Z.Q., Haque, M.M., Binder, K., Sharma, M., Wei, C.-C., and Stuehr, D. J. “Engineering nitric oxide synthase chimeras to function as NO dioxygenases.” J Inorg. Biochem., 158, 122-30, 2016.
- Ding, Y., Sun, D., Wang, G., Yang, H., Xu, H., Chen, J., Xie, Y., Wang, Z.Q. “An efficient PEGylated liposomal nanocarrier containing cell penetrating peptide and pH-sensitive hydrazone bond for enhancing tumor-targeted drug delivery", International Journal of Nanomedicine, 10, 6199-6124, 2015.
- Jensen, D., Reynold, N., Yang, Y.P., Shakya S, Wang, Z.Q., Stuehr, D. J., and Wei, C.-C., “The Exchanged EF-hands in Calmodulin and Troponin C Chimeras Alter the Induced Hydrophobicity and Impair the Interaction with: A Spectroscopic, Thermodynamic and Kinetic Study.” BMC Biochemistry, 16:6, 2015.
- Perera V, Liu, H, Wang, Z.-Q. and Huang, S. “Cell-permeable Au@ZnMoS4 NPs Core-Shell Nanoparticles: Toward a Novel Cellular Copper Detoxifying Drug for Wilson’s Disease”. Chemistry of Materials, 25, 4703-09, 2013.
- Wang, Z.-Q., and Stuehr D. J. “Calcium Signaling: NO Synthase”, In: Lennarz, W.J. and Lane, M.D. (eds.) The Encyclopedia of Biological Chemistry, Vol. 3, pp. 342-346. Waltham, MA: Academic Press, 2013.
- Wang, Z.-Q., Tejero J., Wei, C.-C., Haque, M.M., Santolini J., Fadlalla M., Biswas A., and Stuehr, D. J. “Arg375 tunes tetrahydrobiopterin functions and modulates catalysis by inducible nitric oxide synthase.” J. Inorg. Biochem. 108, 203-15, 2012.
- Tejero, J., Biswas, A., Haque, M.M., Wang, Z.-Q., Hemann, C., Varnado, C.L., Novince, Z., Hille, R., Goodwin. D.C., Stuehr, D.J. “Mesohaem substitution reveals how haem electronic properties can influence the kinetic and catalytic parameters of neuronal NO synthase.” Biochem. J. 433, 163-74, 2011.
- Wang, Z.-Q., Wei, C.-C., and Stuehr, D. J. “How Does a Valine Residue That Modulates Heme-NO Binding Kinetics in Inducible NO Synthase Regulate Enzyme Catalysis” J Inorg. Biochem. 104, 349-56, 2010.
- Wang, Z.-Q., Lawson R. J., Madhavan, B. R., Wei, C.-C., Crane, B. R., Munro, A.W., and Stuehr, D. J. “Bacterial Flavodoxin Support Nitric Oxide Production by Bacillus Subtilis Nitric-Oxide Synthase.” J. Biol. Chem., 282, 2196-202, 2007.
- Wang, Z.-Q., Wei, C.-C., Santolini, J., Koustubh, P., Wang, Q. and Stuehr, D. J. “A Tryptophan That Modulates Tetrahydrobiopterin-Dependent Electron Transfer in Nitric Oxide Synthase Regulates Enzyme Catalysis by Additional Mechanisms.” Biochemistry, 44, 4676-90, 2005.
- Wang, Z.-Q., Wei, C.-C., Sharma, M., Pant, K., Crane, B. R., and Stuehr, D. J. “A Conserved Val to Ile Switch Near the Heme Pocket of Animal and Bacterial Nitric-Oxide Synthases Helps Determine Their Distinct Catalytic Profiles. J. Biol. Chem., 279, 19018-25, 2004.
- Wang, Z.-Q., Wang Y.-H., Wang W.-H., Xue, L.-L., Wu, X.-Z., Xie, Y., and Huang Z.-X., "The Effect of Mutation at Valine-45 on the Stability and Redox Potentials of Trypsin-cleaved Cytochrome b5", Biophysical Chemistry, 83(1), 3-17, 2000.
- Wang, Z.-Q., Wang Y.-H., Wang W.-H., Xie, Y., and Huang Z.-X., "Effect of Mutation at Val45 and Pro40 of Cytochrome b5 on Protein's Stability", J. Inorganic Chemistry, 74, 169, 1999.
- Wang, Z.-Q., Zhang S.-G. Liu, Q.-M., and Ni, J.-Z., "Studies on The Action of Rare Earth with Bovine Serum Albumin in Multimolecular System", Journal of Rare Earth (Special Issue), 573, 1995.
-
Dr. Yaorong Zheng: "Design and development of innovative metal-based chemical systems and their subsequent application in biological systems with a long-term goal of improving cancer therapy"

Dr. Yaorong Zheng
Ph.D. University of Utah, 2011
Associate Professor
236 Integrated Sciences Building
yzheng7@kent.edu
330-672-2267
My research is primarily focused on the design and development of innovative metal-based chemical systems and their subsequent application in biological systems with a long-term goal of improving cancer therapy. Currently, cancer is the second leading cause of death in the United States, only behind cardiovascular disease, and it is projected to become the leading cause of death within 16 years according to American Society of Clinical Oncology. In 2013, approximately 0.6 million U.S. citizens died from cancer, and over 1.6 million U.S. citizens became new cancer patients. The number of new cases is expected to increase nearly 45% by 2030. So far, most types of cancer are still incurable, and development of effective cancer treatments remains at a slow pace. Since 1975, the survival rate for cancer patients has only increased by 3.4%. Several major issues account for the slow development of cancer therapy, including the limited efficacy of current therapeutics and the shortage of efficient drug delivery systems. Research in my lab will focus on developing chemical tools that can benefit cancer research with respect to these limitations. The tools we are working on are based on metal complexes.
Metal complexes play an important role in cancer therapy. Cisplatin, a platinum complex, was demonstrated to be effective in treating cancer in 1969. By virtue of cisplatin, testicular cancer became one of the few types of cancer that are curable, with the survival rate of patients with testicular cancer being greater than 90%. Cisplatin and its analogous, carboplatin and oxaliplatin, are currently FDA-approved anticancer drugs that are widely used in chemotherapy for cancer patients with testicular, ovarian, head and neck, lung, and colon cancer. About 50% of cancer patients with chemotherapy are treated with these platinum drugs. The development of new metal-based drugs is, however, very slow, with most of the research focusing on compounds that are similar to the FDA approved platinum species. Such a drug design strategy favors development of drugs with common intrinsic cytotoxicity and mechanisms to drugs currently in the clinics. Deviating from the traditional approach, we seek to develop novel tools for cancer research based on innovative rationally designed metal-based chemical systems with an emphasis on new applications that target important questions. Our work will focus on applying innovative metal-based chemical systems in combination with nanotechnology and cancer biology to provide new tools to address important issues in cancer research with a long-term goal of improving cancer therapy.
Selected Publications
- Zheng, Y.-R.; Suntharalingam, K.; Johnstone, T. C.; Lippard, S. J. Encapsulation of Pt(IV) Prodrugs within a Pt(II) Cage for Drug Delivery. Chem. Sci. 2015, 6, 1189-1193.
- Zheng, Y.-R.; Suntharalingam, K.; Johnstone, T. C.; Yoo, H.; Lin, W.; Brooks, J. G.; Lippard, S. J. Pt (IV) Prodrugs Designed to Bind Non-Covalently to Human Serum Albumin for Drug Delivery. J. Am. Chem. Soc. 2014, 136, 8790-8798.
- Zheng, Y.-R.; Lan, W.-J.; Wang, M.; Cook, T. R. Stang, P. J. Designed Post-Self-Assembly Structural and Functional Modifications of a Truncated Tetrahedron. J. Am. Chem. Soc. 2011, 133, 17045–17055.
- Zheng, Y.-R.; Zhao, Z; Wang, M.; Ghosh, K.; Pollock, J. B.; Cook, T. R.; Stang, P. J. A Facile Approach toward Multicomponent Supramolecular Structures: Selective Self-Assembly via Charge Separation. J. Am. Chem. Soc. 2010, 132, 16873–16882.
Which Course Should I Register For?
-
Individual Investigation (CHEM 40796)
Individual Investigation (CHEM 40796)
Individual Investigation (CHEM 40796). Most students who wish to begin pursuing a research project will register for Individual Investigation (CHEM 40796). To be eligible, students must have a 2.5 GPA in chemistry (not overall). It is common for students to begin registering for this course in their sophomore or junior year. In this way they will have the opportunity to pursue a project for several semesters prior to graduation which will usually lead to a much more productive and rewarding experience than if a student tries to embark on a project for only one or two semesters in their senior year. There is a form available from the Undergraduate Chemistry Office in 208 Williams Hall that must be completed and signed by your prospective research advisor before you will be allowed to register for the course. Repeat registration is allowed and encouraged; however, while all credit hours will contribute toward your overall 121 credit hour degree requirements, only a limited number of Individual Investigation hours can be used in partial fulfillment of required upper-level major elective hours, as follows: B.S. Chemistry (Chemistry and Materials Chemistry Concentration) and B.A. Chemistry: up to 2 credit hours of CHEM 40796 may be used to fulfill the 2-4 hours of required upper-level CHEM electives; B.S. Chemistry (Biochemistry Concentration): up to 4 credit hours of CHEM 40796 may be used to fulfill the 9 hours of required upper-level CHEM/BSCI electives. A formal written report documenting your research accomplishments must be submitted to your research advisor at the end of the semester, with a copy signed by your advisor forwarded to the Assistant Chair in the Undergraduate Chemistry Office, before a grade can be awarded. An S/U grade is awarded for the course.
-
Senior Honors Thesis (CHEM 40099, HONR 40099)
Senior Honors Thesis (CHEM 40099, HONR 40099)
Senior Honors Thesis (CHEM 40099, HONR 40099). Students who are planning to graduate with University Honors typically pursue undergraduate research leading to the writing of a Senior Honors Thesis. Qualified students who have not completed all the requirements for graduation with University Honors can still graduate with Departmental Honors by completing a Senior Thesis. Usually, a student will register for Individual Investigation until the summer of their junior year. During the following year, they would register for between 5 and 10 semester hours of Senior Honors Thesis under either CHEM 40099 or HONR 40099. Departmental and Honors College approval are required prior to undertaking such thesis work. Towards the end of the final semester, a thesis describing the research work must be written and defended before a small faculty committee. This is an excellent opportunity to learn how to write and defend a scientific thesis, skills which will prove invaluable in graduate school or industry.
Time Commitment
Research schedules are set with your advisor and vary by lab.
As a general guideline, expect to spend:
At least 4 hours per week (about 60 hours per semester) for each credit hour.
You’ll typically have set lab times each week, but some weeks may require additional time depending on your experiments.
